Patients with hypermobility spectrum disorder (HSD) or hypermobile Ehlers-Danlos Syndrome frequently also have problems with their gut. Symptoms can include but are not limited to gastric reflux (“GERD”), heartburn, constipation, diarrhea, belching, bloating, cramps, nausea, difficulty swallowing, abdominal pain, cramping, difficulty swallowing, feelings of fullness. One author wrote about gastrointestinal (GI) problems in HSD/hEDS as follows:
“GI manifestations should be considered major contributors to disability in JHS/EDS-HT [joint hypermobility syndrome/ Ehlers-Danlos Syndrome Hypermobility Type]. However, the scarce knowledge of their pathophysiologic basis explains why surgery and standard pharmacologic treatments are usually of minor effect at the long-term, with great frustration for patients and practitioners.” {3, pg. 68}
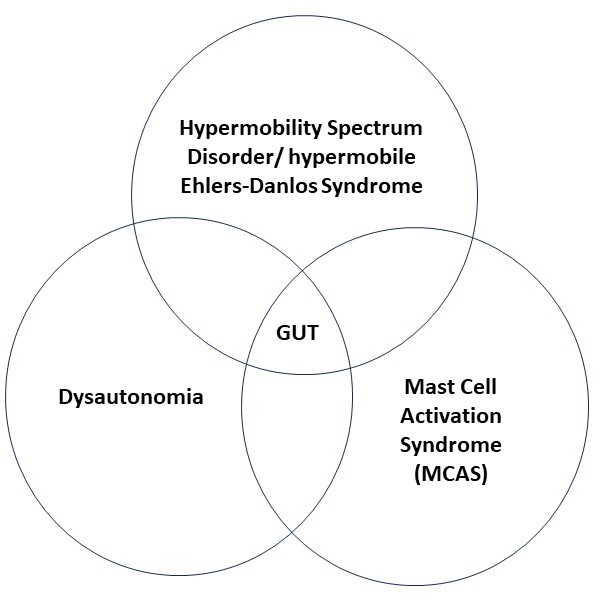
HSD.hEDS/ POTS/ MCAS and the Gut {1, 11}
In past blogs, we have talked of the connection between hypermobility and dysautonomia (which may or may not include postural orthostatic tachycardia syndrome, POTS) and mast cell activation syndrome (MCAS). While these three syndromes are recognized by many clinicians as occurring with increased frequency together, both the real frequency of these associations and their reasons have not yet been made clear in the medical literature. Even so, the potential overlap between these three syndromes can provide a useful model for understanding some of the issues patients face with their gut.
Weak or overly stretchy connective tissue in the gut can lead to structural problems as a result of HSD/ hEDS.
The imbalance of the involuntary nervous system which is dysautonomia can lead to problems with nervous system governed movements of the gut.
And MCAS results in inflammation which can also lead to many symptoms.
As you can see, the effects of each of these syndromes can overlap with each other and can all meet to cause gastrointestinal, or more easily said – gut, problems. While not all gut problems encountered by hypermobile patients fit neatly into this structure-nervous system-inflammatory reference, we will start with it as an organizing principle here because it does assist understanding.
Structural Problems {3, 4}
The connective tissue changes of HSD or hEDS may result in a number of structural problems in the gastrointestinal tract. One of these problems can be altered compliance, stretchiness, of the gut wall. There should normally be a certain amount of tension, or recoil, in the gut wall. This assists the movement of food through the gut tube in the process called peristalsis. Changes to the gut wall can be one of the factors involved with sluggish peristalsis which when severe leads to gastroparesis and constipation.
The gut wall has a lot of arteries feeding it and veins taking blood away. Connective tissue structural changes can alter the tensions in the blood vessels which help keep the blood flowing smoothly. Blood can pool and not be efficiently removed by the veins causing symptoms.
While not common, weakened connective tissue in the gut wall and blood vessels can lead to hemorrhages or perforations.
Hernias in which the gut pushes out through a weak area in the abdominal wall (abdominal hernia) or in the breathing diaphragm (hiatal) are particularly common in HSD and hEDS.
The organs in the abdomen, including the uterus plus small and large intestines, are held in place by a number of ligaments, which are of course made of connective tissue. If these sag or stretch, the intestines will no longer sit in their normal position. This is called organ ptosis and can lead to pinching or kinking or sluggish blood flow for that organ. In severe cases, a prolapse may occur where the organ drops down far out of its normal position. Examples of two prolapses are the rectum coming out of the anus or the uterus protruding from the vagina.
There can be other structural problems of the gut in HSD and hEDS, but these are the main ones which have been documented in the literature so far.
Inflammatory Problems {2, 7, 10, 14}
Three inflammatory bowel diagnoses in which the occurrence of hypermobility are increased are irritable bowel disease (IBS), Crohn’s Disease and ulcerative colitis. {2} IBS is the more generic diagnosis with Crohn’s disease and ulcerative colitis being more specific sub diagnoses. While these disorders are listed as inflammatory disorders, there are also significant contributions to them from the immune system and the nervous system. In fact, while all of these overlapping diagnoses are inflammatory, the cause is unknown and thought to involve a number of factors including intestinal bacteria, genetic and environmental causes. {14}
While the causes of IBS are multifactorial, there is evidence that in a number of patients with treatment resistant IBS there is an increased number of mast cells as well as mast cell activity in the gut. {7} This points to the overlap with MCAS.
Nervous System Problems {5, 8, 9, 10, 15}
Food being digested by the gastrointestinal tract is pushed forward in the small and large intestines by contractions of muscles in the gut wall. These contractions are largely affected by the involuntary nervous system. When these contractions are impaired in their strength or speed of contraction, this is called dysmotility. Dysmotility has been found to be common in patients with HSD/hEDS. If the contractions are severely slowed down or stopped, this is gastroparesis which results in constipation, and constipation is a common complaint among hypermobile patients.
Another common complaint among patients with hypermobility is abdominal pain. This may be mild to severe and in worst cases profoundly affect the patient’s life. As for the body as a whole, pain is a perception created by the brain based on its best estimate that the body is being hurt. Local nerves in the gut send signals to the spinal cord which filters them and passes on the strongest and most urgent. When these signals get to the brain, they are interpreted by a complex interaction of a number of brain areas. The precise combination and contribution of the different brain areas is unique to every individual person and is called a pain signature. Scientists who study pain have come to believe that in many cases of chronic or recurring pain, the nervous system has become sensitized and overly efficient at creating pain. This is called visceral hypersensitivity.
This sensitization can happen at the nerves in the gut, in the spinal cord, in the brain, or in any combination of these. When the nerves become sensitized, then the nervous system becomes as much or more a part of the pain problem than any local disease or dysfunction. Because the nervous system is in great part a learning organ, many things can contribute to making the brain’s damage estimate too strong. How long the problem has been going on, anxiety, depression, history of physical or sexual abuse especially in childhood, past gut disease can all be major factors in the nervous system developing visceral hypersensitivity.
Disorders of Function {2, 3, 7, 12}
Many of the gut problems which affect people with HSD/ hEDS fall in the category of “functional” problems. This means that they are not predominantly structural or neurologic or inflammatory but have overlapping physical causations.
Because food sensitivities and allergies are often a part of mast cell activation syndrome, some people are very challenged to maintain a balanced, gut -friendly diet. A balanced diet requires the right combination of protein, low-sugar carbohydrates and fats. The challenges of finding a diet that does not worsen symptoms such as diarrhea, constipation, bloating, gas, cramping can lead to other problems as well as nutritional imbalances.
GERD is gastroesophageal reflux disease. This means that stomach acids wash up, or reflux, into the lower part of the esophagus causing irritation and pain and “heart burn”.
An inflamed gut, which occurs in Crohn’s disease, irritable bowel syndrome and ulcerative colitis may lead to a “leaky gut” in which bacteria or tiny food particles passing through the gut can pass between the normally tight junctions between the gut cells and access the blood. This can lead to many symptoms including immune system reactions. Leaky gut is not recognized by all physicians, but according to the gastroenterology literature us most likely in an inflamed bowel.
Small intestinal bacterial overgrowth (SIBO) and its cousin small intestinal fungal overgrowth (SIFO) occur more frequently in hypermobile patients with gut problems. The reason for this is not clear, but these overgrowths can lead to symptoms such as belching, bloating, abdominal pain, diarrhea, flatulence, and indigestion.
Gluten sensitivity, and it’s more severe form Celiac disease, are more common in patients with hypermobility.
Finally, medications can have a strong effect on the gut. Opiates are famous for causing painful constipation. Patients with hypermobility, especially those with MCAS, may be sensitive to or even allergic to some medications. And being on multiple medications may cause drug interactions or have conflicting effects on the body (This is sometimes called “Poly pharmacy” in the medical community.)
In the next post in this short series, we will explore treatment approaches for the gut problems we have talked about here.
Until then – cheers!
Zeborah Dazzle, PT, WWF and Mark Melecki, PT, DPT, OCS

References:
- Wong, S., et. al, The Gastrointestinal Effects Amongst Ehlers-Danlos Syndrome, Mast Cell Activation Syndrome and Postural Orthostatic Tachycardia Syndrome. AIMS Allergy and Immunology, 6(2): 19-24, DOI: 10.3934/Allergy.2022004
- Beckers, A.B., et. al., Gastrointestinal Disorders in Joint Hypermobility Syndrome/ Ehlers-Danlos Syndrome Hypermobility Type: A Review for the Gastroenterologist. Neurogastroenterology & Motility 2017; 29:e13013: 1-10; doi.org/10.1111/nmo.13013
- Castori, M., et. al., Gastrointestinal and Nutritional Issues in Joint Hypermobility Syndrome/ Ehlers-Danlos Syndrome, Hypermobility Type. American Journal of Medical Genetics Part C (Seminars in Medical Genetics) 169C: 54-75 (2015); doi 10.1002/ajmg.c.31431.
- Thwaites, P, et. al., Hypermobile Ehlers-Danlos Syndrome and Disorders of the Gastrointestinal Tract: What the Gastroenterologist Needs to Know. Journal of Gastroenterology and Hepatology 37 (2022) 1693-1709; doi:10.1111/jgh.15927
- Farmer, A.D. & Aziz, Q., Visceral Pain Hypersensitivity in Functional Gastrointestinal Disorders. British Medical bulletin 2009; 91: 123-136; doi:10.1093/bmb/ldp026.
- Camilleri, M., The Leaky Gut: Mechanisms, Measurement and Clinical Implications in Humans. Gut 2019 August; 68(8): 1516-1526. Doi:10.1136/gutjnl-2019-318427
- Freiling, T., et. al., Evidence for Mast Cell Activation in Patients with Therapy Resistant Irritable Bowel Disease. Z. Gastroenterol 2011; 49:191-194. http://dx.doi.org/10.1055/s-0029-1245707.
- Gottfried-Blackmore, A., et. al., Open Label Pilot Study: Non-Invasive Vagal Nerve Stimulation Improves Symptoms and Gastric Emptying in Patients with Idiopathic Gastroparesis. Neurogastroenterol Motil. 2020 April; 32(4): e13769.doi:10.1111/nmo.13769
- Frokjaer, J.B., et. al., Modulation of Vagal Tone enhances Gastroduodenal Motility and Reduces Somatic Pain Sensitivity. Neurogastroenterol Motil (2016) 28, 592-598; doi:10.1111/nmo.12760
- Cirillo, G., et. al., Vagus Nerve Stimulation: A Personalized Therapeutic Approach for Crohn’s and Other Inflammatory Bowel Disease. Cells 2022, 11, 4103; https://doi.org/10.3390/cells11244103
- Kohn, a., Chang, C., The Relationship Between Hypermobile Ehlers-Danlos Syndrome (hEDS), Postural Orthostatic Tachycardia Syndrome (POTS), and Mast Cell Activation Syndrome (MCAS). Clinical Reviews in Allergy & Immunology (2020) 58: 273-297; https://doi.org/10.1007/s12016-019-08755-8.
- Uy, P., et. al, SIBO and SIFO Prevalence in Patients with Ehlers-Danlos Syndrome Based on duodenal Aspirates/ Culture. The American Journal of Gastroenterology 116, PS218-S219, October 2021; doi:10.14309/01.ajg.0000774444.60980.27
- Danese, C., et. al., Screening for Celiac Disease in Joint Hypermobility Syndrome/ Ehlers-Danlos Syndrome Hypermobility Type. American Journal of Medical Genetics Part A 9999:1-3.
- Torres, J., et. al., Crohn’s Disease. Lancet 2017; 389: 1741-1755; http://dx.doi.org/10.1016/S0140-6736(16)31711-1.
- Alomari, M., et. al., Prevalence and Predictors of Gastrointestinal Dysmotility in Patients with Hypermobile Ehlers-Danlos Syndrome: A Tertiary Care Center Experience. Cureus, 2020, 12(4): e7881; doi.10.7794/cureus.7881
